By Phil Byrne, COMSOL Applications Engineer, Stockholm, Sweden
It’s been reported that the global scientific community concurs that, for long-term management, it is acceptable and safe to dispose of spent nuclear rods in deep geological respositories. Many sites throughout the world are being considered as repositories.
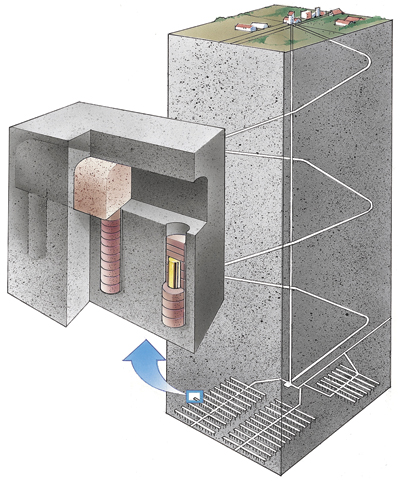
The repositories typically consist of two important containment barriers. One is a container that protects the waste and prevents any water from reaching it for hundreds of thousands of years. It is made of copper and is specially welded and treated. The other barrier is the bentonite buffer material that protects the containers and prevents water from flowing through to them, thereby mitigating any deep-earth movement, and binding any radionuclides that eventually escape from the containers.
Bentonite is used in the drilling and mining industries due to its unique rheological properties. It is a naturally occurring type of clay that expands when it comes in contact with water. This material allows almost no water to flow through it. Expanding bentonite fills the space surrounding the final disposal canisters. In the event of a possible canister leak, bentonite retards the radioactive substances from coming into contact with the rock through its resistance to radionuclide diffusion.
Research groups are studying the effectiveness of bentonite as a containment barrier. These activities consist of work with mathematical models, since testing is extremely difficult due to the time involved. The Swedish Nuclear Fuel and Waste Management Company (SKB) and Finland’s equivalent company Posiva Oy have collaborated with several research groups in Sweden and Finland to model and study the repositories. These include a group led by Docent Markus Olin at VTT Technical Research Centre of Finland (VTT) and a group led by Professor Ivars Neretnieks and Dr. Luis Moreno at the Division of Chemical Engineering at the Royal Institute of Technology, Sweden (KTH).
Olin and his colleagues are investigating what happens during the excavation of the tunnels and placement of the canisters (days to months), the saturation of bentonite by water (months to hundreds of years), and the long-term safety where breaches can occur such that the surrounding system will have to retard radioactive material reaching the biosphere (many thousands of years). They are also considering different but interacting physical phenomena that influence the repositories; hydrological, thermal, mechanical, and chemical phenomena.
Veli-Matti Pulkkanen at VTT produced 3D models over the whole bentonite and surrounding rock including fractures that cut through it where a hypothetical leak of radionuclides from a canister is simulated. When the tunnels and holes are excavated, a thin layer of bedrock surrounding them may be damaged known as the excavation damaged zone (EDZ), and it is easier for water to flow through the small pores and fractures that have resulted from this excavation. It may be easier for fluid to flow in the EDZ than through the original tunnel.
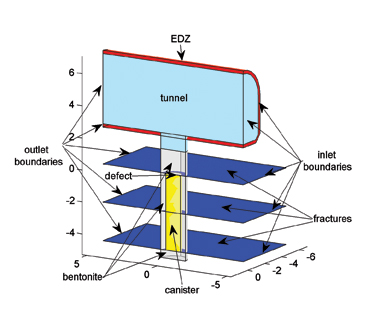
Pulkkanen uses COMSOL Multiphysics software to define the source of radionuclide leaking from the canister. He uses a simple 1D initial boundary-value problem, and couples it to the natural 3D geometry. The same technique is also used to define the flow physics in 2D for thin fractures and 3D flow. This ability to couple physics defined in different dimensions comes as an automatic interface in the software and requires only specifying the relevant material properties — the coupling between the different geometries is done automatically and the meshing is said to be simple to manipulate. His results indicate that the bentonite layer is an important hindrance to the transport of leaking radionuclides.

Olin simulated the chemical stability of bentonite using the Reaction Engineering Module. The susceptibility of bentonite to dissolve in relatively low-saline water, which could reach the repositories after post-glacial periods, is greater if its makeup is dominated by sodium ions versus calcium ions. Even if the bentonite is in contact with saline water, the exchange of calcium ions with sodium ions can also occur.
Olin defined and solved three chemical equations and their corresponding non-linear mass-action laws, three mass balance equations, two charge balance equations, and an activity coefficient model. From this simplest possible chemical model of bentonite, he determined the equivalents of the two ions in the bentonite as a function of the equivalent of calcium ions in water, and showed that the relationship is nonlinear. Olin hopes to incorporate his reaction model into a model of transport between a fracture and the bentonite and the structural behavior of saturated and swelling bentonite.
The group led by Neretnieks is investigating how the structural properties of bentonite affect its dissolution. More research groups and institutions will turn to modeling to adequately simulate the behavior of their nuclear waste and repositories. The time-scales associated with the behavior make experiments and testing rather limited, so that highly accurate estimations of the future consequences are required.
COMSOL
www.comsol.com


Leave a Reply
You must be logged in to post a comment.