Medical device manufacturers are motivated to deliver groundbreaking drug delivery solutions driven by new pharmaceutical therapies and smart connected products that have the potential to transform the patient care paradigm from in-patient care to outpatient and self-administered therapies. Innovators that provide a compelling and intuitive user experience have a competitive advantage. However, medical device makers face challenges in their quests to develop innovative devices that meet regulatory requirements, achieve sustainability goals and improve patient experience and outcomes.
To address these challenges, medical device makers are leveraging computer-aided design (CAD), modeling, and simulation technologies unified on a collaborative and data-managed platform. This unified modeling and simulation (MODSIM) approach from Dassault Systèmes enables scientists, researchers, engineers and designers to collaboratively develop and evaluate pharmaceutical therapies combined with mechanical and electronic devices for optimal patient care.
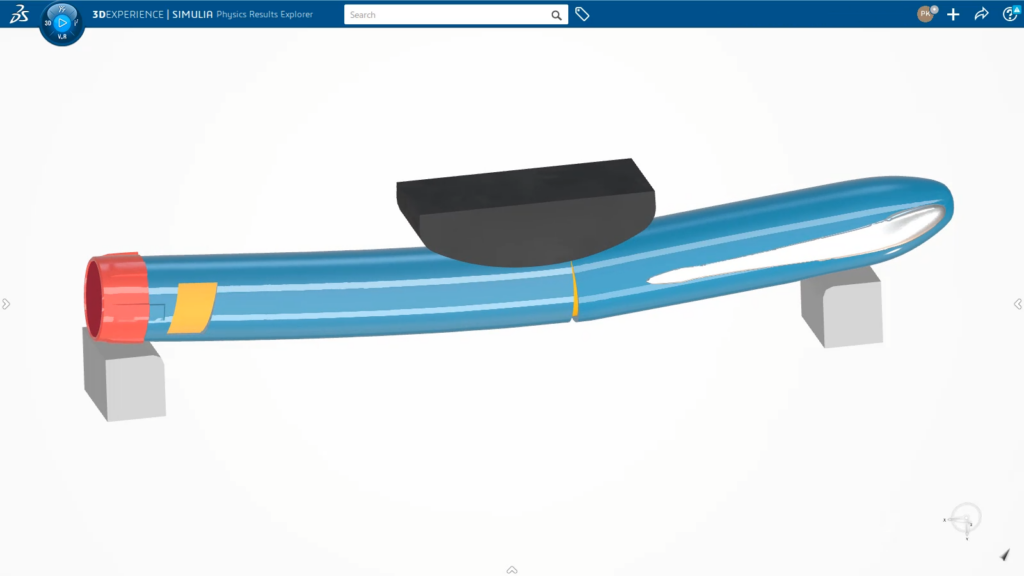
MODSIM delivers full associativity between CAD and computer-aided engineering (CAE) models and connectivity with functional requirements, product designs and simulation results to ensure decision traceability. Beginning with conceptual designs, designers and engineers can collaborate across work functions on developing a device and its components tailored to delivering a specific drug therapy. They can analyze a range of physics that influence design performance, including structural analysis of components and assemblies, fluid dynamics and fluid-structure interactions of the drug delivered through the device and electromagnetic analysis of the electronic components and antennas for wireless connectivity.
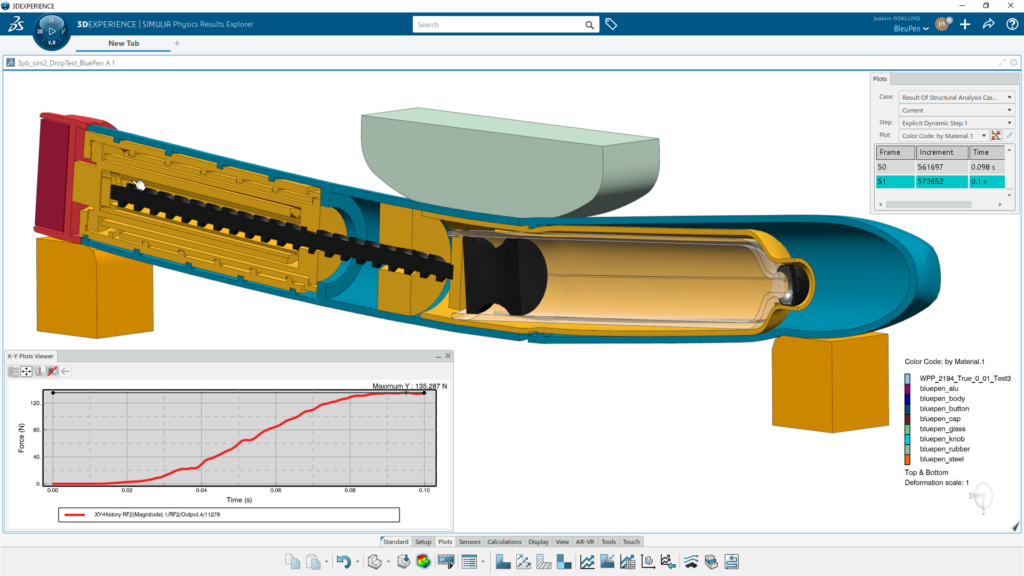
Combined with design exploration, optimization and data management applications, MODSIM improves upon basic CAD and CAE integration. MODSIM enables workflow automation to eliminate laborious, repetitive and error-prone tasks such as updating simulation models and re-meshing each design revision. Best practices for modeling and simulation are easily captured, templatized and distributed to non-experts so they can reliably assess the implication of design modifications. Data management apps and the cloud platform allow them to share their simulation results and share their thinking on performance-based decisions, regardless of their physical location.
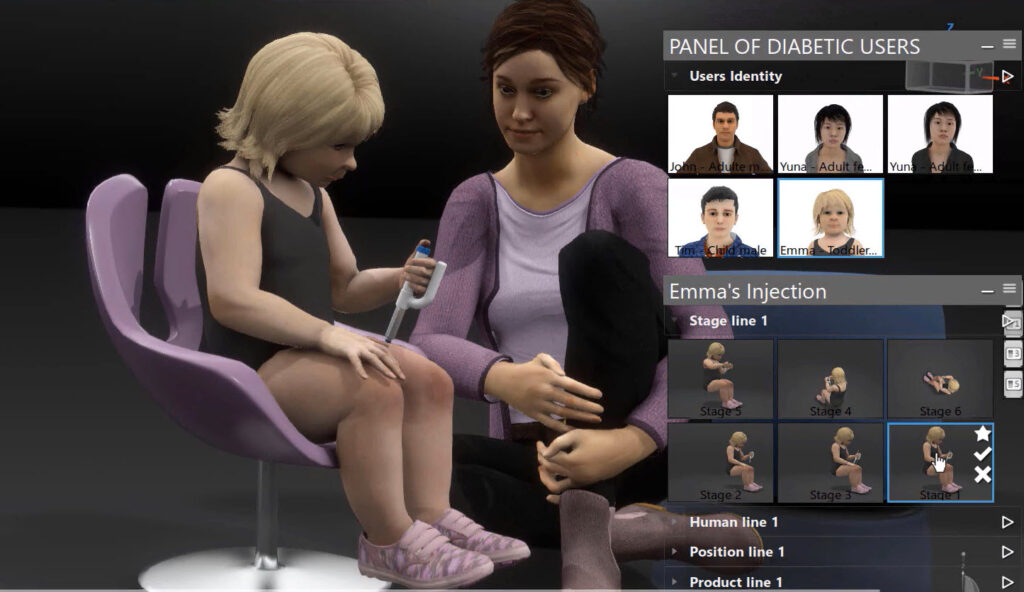
Beyond the device function itself, developers need to evaluate the physical and physiological interaction of medical devices with the human body. For instance, when developing a drug delivery device, designers can use a virtual twin of the human body, which is a dynamic virtual 3D model, to analyze the sizing and ergonomics of their device, as well as the force required to insert a needle into the skin for a variety of patient types ranging from adults to children. Similarly, cardiac implant makers can use a virtual twin of the heart to evaluate how an artificial heart valve will move during the cardiac cycle in a virtual environment before physical prototyping or testing on humans. Significant technical advances are being made in the development of realistic, validated virtual models of human organs and musculoskeletal systems to enable the assessment of medical device safety and effectiveness.
Realistic modeling and simulation of medical devices interacting with virtual humans offers the promise of creating personalized virtual twins to evaluate the individual patient experience, device safety and therapeutic outcomes.
Dassault Systèmes Life Sciences & Healthcare
www.3ds.com/industries/life-sciences-healthcare


Leave a Reply
You must be logged in to post a comment.